Now what we need to do is figure out about the ion itself. We're going to leave myself a note next to potassium, that says big. Most of the time we know that we can expect chlorine to be larger than Potassium. As you go down a group, you are Adam's are getting bigger as the radius gets larger. When you're going across a row on the periodic table, your Adam's tend to be getting smaller, which means they have smaller radio. The general trends in size are the first thing we'll talk about. (Figure 1)/the picture connected to this question Suppose that each sphere represents 5.1×10 21 molecules and that the volume of the container is 1.0 L. 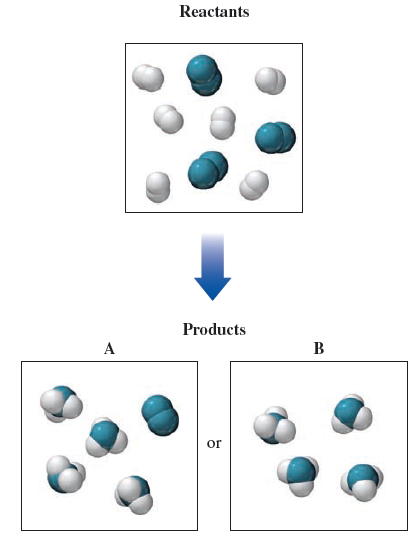
They are separated in terms of the atomic number. The following pictures represent the progress of the reaction AB in which A molecules (red spheres) are converted to B molecules (blue spheres). You can see where the atoms are positioned by looking at where chlorine is in the next to last column on Row three, and then potassium is in the first column or first group on the period. The location of the atoms will tell us a lot about their size and radius, so it's important that you have a periodic table nearby. The larger Adam's radius is, the more problems he will face. The size of the atom is what we're talking about when we talk about radius. Understand what it means to have a radius. There are three options for their radius. We've been given three different atoms, one of which is a positive ion and the other two negative islands. We get a question about the atomic radius. Hot coffee: Hot coffee is typically prepared by steeping ground coffee in hot water and then removing the spent coffee grounds.All right. Carbon monoxide: Carbon monoxide has the formula. Soil: Soil contains air, minerals, water, and organic matter. Orange juice with pulp: Orange juice with pulp contains liquid juice, which is mostly water, and some of the fruit solids (pulp). Salt water: Salt water is water that contains dissolved salt. The gases are not chemically bonded to each other. In chemistry, the ball-and-stick model is a molecular model of a chemical substance which displays both the three-dimensional position of the atoms and the bonds between them. Explanation of the different examples of matter The eleven examples of matter in this item are the following: Air: Air is composed of about 78 nitrogen and 21 oxygen, with traces of argon, carbon dioxide, and other gases. Classify each type of matter as a pure substance or a mixture Classify each type of matter as a pure substance or a mixture. Match the words in the left-hand column with the appropriate blank in the sentences in the right-hand column. In the following drawings, red and blue spheres represent atoms of different elements. Define the terms Complete each sentence with the appropriate term. Choose the homogeneous mixture from the list below. It may help to use the periodic table to determine whether a substance is an element. The white and blue spheres represent boron and nitrogen atoms, respectively, the orange isosurface is the hole polaron in a single boron nitride layer that is being exfoliated. Then, further classify each pure substance as either an element or a compound, and classify each mixture as either a heterogeneous mixture or a homogeneous mixture. In addition, mixtures do not have constant composition. A mixture, in contrast, is a combination of substances in which each substance retains its own identity. A pure substance has a specific chemical composition, and all samples of the How to approach the problem First, identify each type of matter as a pure substance or a mixture. State Atomic/molecular motion Atomic/molecular spacing Shape Volume Solid oscillation/vibration about fixed point close together definite definite Liquid free to move relative to one another close together indefinite definite Gas free to move relative to one another far apart indefinite indefinite ANSWER: Correct Part B Classify each type of matter as an element, a compound, a heterogeneous mixture, or a homogeneous mixture. The properties of solids, liquids, and gases The following chart describes some of the properties of solids, liquids, and gases. What state of matter is depicted in each bin? Drag each item to the appropriate bin. Part A The blue spheres below represent atoms. Matter is classified in many different ways, including by its state (solid, liquid, or gas) and by its composition (pure substances or mixtures). Classifying Matter Matter is anything that takes up space and has mass. CH 03 Matter & Energy Due: 11:59pm on Tuesday, FebruTo understand how points are awarded, read the Grading Policy for this assignment. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
